An advanced treatment for patients who have not found relief
for their depression and anxiety
TMS has been recommended by the American Psychiatric Association since 2010 and FDA cleared since 2008 for the treatment of major depressive disorder.
TMS is free from common antidepressant drug side effects such as weight gain and sexual dysfunction. TMS is not to be confused with ECT, and does not affect cognitive function such as memory.
TMS is an innovative, non-drug therapy that helps people who struggle with Major Depressive Disorder. It helps activate the natural function of the brain’s neurotransmitters using a non-invasive magnetic field.
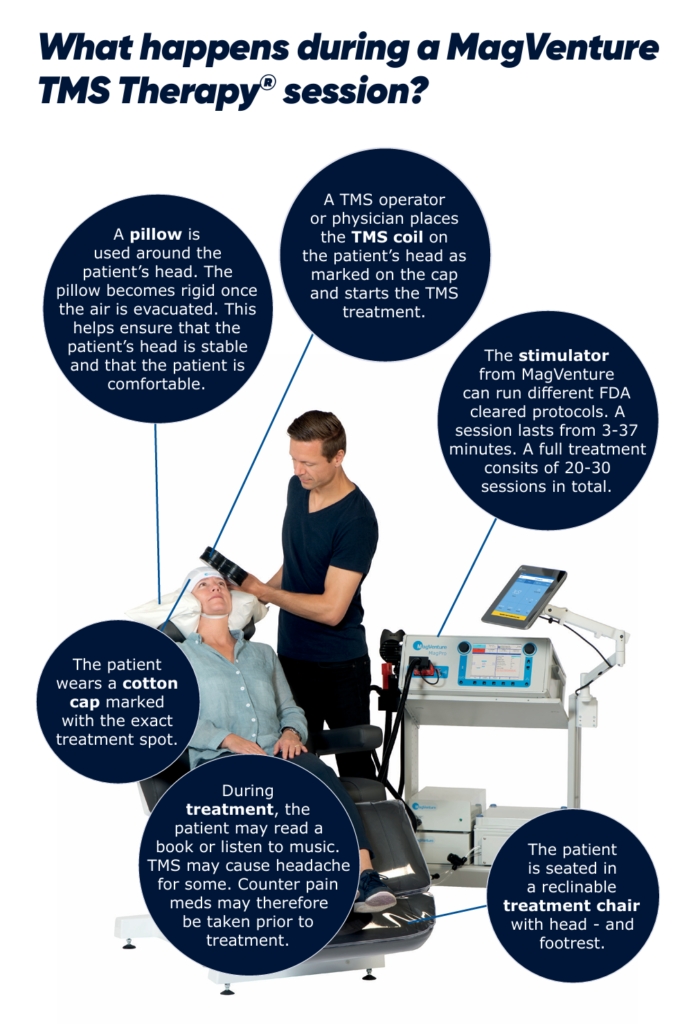
Administered in our office without sedation, TMS requires 36 treatments. Each treatment takes approximately 20 minutes and is administered five days per week. Magnetic pulses are used to stimulate specific mood centers of the brain. We also offer another form of TMS, Thetaburst, which requires a 3-minute treatment.
Hear Ann Wycoff, Ph.D. explain the benefits of TMS for treating depression and anxiety.
Hear Dr. Bruce Hubbard, M.D. explain the benefits of TMS for treating depression and anxiety.
TMS is a breakthrough in the treatment of major depressive disorder because it offers real potential for long-term remission of symptoms. With standard drug therapy, long-term remission becomes less likely with each prescription medication treatment attempt. As antidepressant medication increases, so can side effects and the potential for treatment failure.
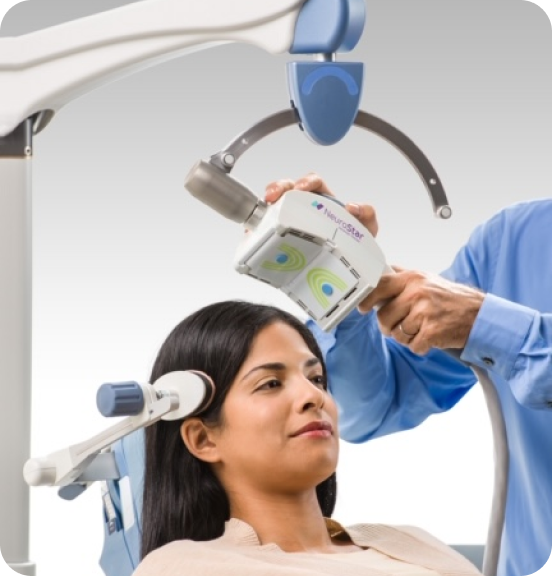
TMS uses a targeted pulsed magnetic field, similar to what is used in an MRI (magnetic resonance imaging) machine. While the patient is awake and alert, magnetic pulses are delivered to stimulate nerve cells in the part of the brain controlling the mood which is often underactive in patients with depression.
When getting TMS, you sit in a chair in a relaxed position. When the magnetic pulses are delivered you hear a clicking sound and feel a tapping sensation on your head. Most patients are able to watch TV or chat with their treater during the session. There are no restrictions to you activity follow a treatment. You can leave our office, drive, and return to you daily activities.
Powered By EmbedPress
Consider these questions and contact our office to discuss whether TMS Therapy is right for you:

TMS can be used to treat symptoms other than Depression by targeting different areas of the brain. Research has shown that TMS can be helpful in reducing anxiety where other treatments have failed. A brain without anxiety can regulate emotions, impulses, and behaviors by controlling the limbic system—the emotional center of the brain. If you suffer from an anxiety condition, like Generalized Anxiety Disorder (GAD), the part of your brain called the amygdala is hyperactive. TMS treatment can reduce activity in the amygdala thus reducing your symptoms and allowing you to a live more calmly.
Studies have also shown that TMS can help reduce symptoms of Obsessive-Compulsive Disorder (OCD) such as recurring or intrusive thoughts and ritualistic behavior. People that suffer from OCD often have symptoms of depression as well, so our TMS treatment protocols involve treating both the depression and OCD symptoms concurrently.
Click on TMS for OCD to learn more about this exciting new treatment for the intrusive thoughts found in OCD.
Contact our office to arrange a consultation to determine if TMS is right for you.

“I spent 22 years living with depression and now I have full range of emotions that I’ve never experienced before, and a new outlook on life. Before beginning treatment, I was non-functioning and almost completely non-verbal. My sleep schedule was non-existent, my energy levels were very low, and I had to pause my schooling because I was unable to even talk to people.
By week two of treatment, not only was I sleeping better and waking up refreshed, but those around me noticed a significant change. I began talking so much I joked that my therapist would have to teach me to be quiet. This treatment has changed my life for the better. I would highly recommend TMS to anyone suffering from severe depression.“
“I noticed immediately an improvement and so did the people around me. That heavy weight of depression was lifted and I felt good. I’m happier than I’ve been in years. The medicines weren’t working so I got proactive and tried this. I’m so patient with my husband now. I don’t have that anger anymore. I laugh every day many times and that’s really new. I’ve never been to a place with so much empathy and compassion. I felt I could let down and show it all. The staff here is deeply invested in their patients. What I experience now is positivity. I guess that’s the right word. My husband is so happy with the results. I didn’t smile for so long. I don’t have the moodiness. There are so many depressed people in the world that this will help. If anyone wants to email me or talk with me I’d be happy to talk with them.”
Scholarly Article: Repetitive Transcranial Magnetic Stimulation (rTMS) in Treatment Resistant Depression: Retrospective Data Analysis from Clinical Practice.
Scholarly Article: Has repetitive transcranial magnetic stimulation (rTMS) treatment for depression improved? A systematic review and meta-analysis comparing the recent vs. the earlier rTMS studies.
Scholarly Article: Efficacy and Safety of Transcranial Magnetic
Stimulation in the Acute Treatment of Major
Depression: A Multisite Randomized Controlled Trial.
Scholarly Article: Transcranial magnetic stimulation (TMS) in the treatment of attention-deficit/hyperactivity disorder in adolescents and young adults: a pilot study.
Scholarly Article: Clinical Significance of Transcranial Magnetic Stimulation (TMS) in the Treatment of Pharmacoresistant Depression: Synthesis of Recent Data.
Scholarly Article: Transcranial magnetic stimulation for the treatment of major depression.
TMS therapy is well tolerated and has been proven to be safe in clinical trials. The most commonly reported side effects related to treatment were scalp pain or discomfort during treatment sessions. These side effects were generally mild to moderate and occurred less frequently after the first week of treatment.
Our services are covered by most commercial health insurance plans in addition to Medicare and Tricare. Please have your health insurance information at hand when you call to schedule an initial evaluation. Our TMS Coordinator will check your benefits and assist you in requesting prior authorization.
Results have persisted for at least one year for most subjects followed by researchers. Many patients report results lasting several years.
Anyone who is currently depressed may be a candidate for TMS. TMS is especially beneficial for those who have not adequately benefited from antidepressant medications, or who cannot tolerate the side effects of these medications.
The research shows that a small number of patients may experience a mild, dull headache either during or right after treatment. It subsides within an hour or two. We rarely see this side effect but recommend that the patient take Tylenol before treatment if they experience this. We have only seen this side effect in the first week of treatment, and we rarely see it at all. There is only one other “side effect” that we have seen in our experience in administering TMS to over 400 patients. About 50% of patients will experience an “emotional dip” at some time during their treatment. This usually occurs at about the third week and consists of a temporary return to the level of depression they felt when they first started treatment. The “dip” usually lasts for 2-5 days. In rare circumstances it can last up to two weeks. It appears to be correlated with the presence of past trauma. Many patients do not experience a dip at all. It is believed by some experts that when we stimulate the prefrontal cortex, repressed feelings that are stored in that area of the brain are also stimulated, causing those old, painful feelings to surface. If a dip occurs, we watch the patient closely and provide extra support during this period. The patient schedules in-person check-ins with Dr. Wycoff as often as needed. Dr. Wycoff stays in close contact with the technician delivering the daily treatment and with Dr. Hubbard, our psychiatrist. Dr Hubbard will meet with the patient in-person if indicated. When the patient is reminded that this is a normal and expected part of treatment, they tend to relax and feel better. The dip will gradually subside and it is at this point that the effects of TMS tend to gain momentum. An emotional dip is a sign that the treatment is working. Although the “dip” is unpleasant it can be an indicator of a significantly positive result by the end of treatment.
Yes. We do not want the patient to make any substantive changes to their regular medication routine while undergoing TMS. After the patient has completed TMS they can discuss with their provider whether it is appropriate to gradually discontinue medication. Patients should never stop their medication without the supervision of a medical provider, as abrupt discontinuation of an antidepressant may have serious adverse effects.
Yes, TMS treatment is noninvasive and the patient is not sedated or medicated for the treatment. After treatment the patient drives home and continues their normal routine.
TMS is FDA approved for the treatment of depression. There are twelve years of solid, high quality, peer reviewed research supporting TMS. We have treated over 400 patients and have not seen any harmful effects to any of our patients. The majority of patients experience a significant reduction in depression which, according to the research and our own experience, lasts for at least one year but much longer for many.
According to the research and our experience, younger patients tend to get an earlier effect. Patients in their 20’s often feel an effect by the 3rd or 4th treatment which gradually gains momentum. Patients in their 40’s and 50’s typically get an effect after 2-3 weeks. Older patients may not feel an effect until the 4th week of treatment. If a patient only begins to get a solid effect much later in the treatment protocol, we will often ask their insurance to give them ten more treatments. If their insurance denies our request, we will often give ten extra treatments at no charge. We have seen patients benefit significantly from ten more treatments if they only begin to feel a significant effect later in the protocol. Our goal is to pay close attention to each patient’s individual experience and adjust treatment when needed to get each patient the very best result possible.
Yes. Depression and anxiety frequently co-exist. The typical TMS protocol delivers a magnetic pulse to the prefrontal cortex on the top left side of the head typically reduces both depression and anxiety symptoms together. However, some patients are experiencing severe anxiety in addition to depression. For those patients we may treat the top right side of the head in addition to the left side treatment.
Choosing the right provider is important for treatment success. Learn what to look for when evaluating your options: